Surfactant selection is one of the most consequential decisions in a cleansing formulation. The class of cosmetic surfactants you choose determines foam volume and texture, skin feel after rinse, compatibility with active ingredients, and the claims you can defensibly make at launch. Get the combination right, and the formula performs across all three dimensions: consumer perception, skin compatibility, and stability. This guide covers the four ionic classes, amino acid-derived alternatives, and the key selection trade-offs that matter most when building a real-world cleansing system.
The scale of the category reflects how foundational surfactants are to beauty and personal care product development. The global beauty and personal care surfactants market was valued at $11.69 billion in 2025, and the personal care surfactants segment is projected to grow from $8.9 billion in 2026 to $13.8 billion by 2036 at a 4.5% CAGR. Much of that growth is being shaped by demand for milder, sulfate-free, and amino acid-based systems, trends that make surfactant class selection more consequential, not less.
Key Takeaways
- The four primary ionic classes of cosmetic surfactants (anionic, cationic, amphoteric, and nonionic) each serve distinct functional roles; most finished cleansing formulas use two or more classes in combination.
- Anionic surfactants deliver the highest foam and detergency but carry the greatest potential for barrier disruption and irritation; amphoteric co-surfactants are routinely added to moderate that risk.
- Amino acid-derived surfactants (glutamates, sarcosinates, isethionates) offer a milder skin compatibility profile than traditional anionics, making them the preferred primary for premium, sensitive-skin, and baby care positioning.
- Sulfate-free systems require a viscosity strategy beyond surfactant substitution: standard NaCl salt thickening loses effectiveness without SLS/SLES, and an alternative rheology approach is essential.
- Surfactant selection should be driven by product category, target skin type, foam expectation, pH operating range, and compatibility with any actives present in the formula.
- Vivify Beauty Care’s surfactants and cleansing agents portfolio includes options across these classes, with filtering by certification, physical form, and product type suitability.
What Are Cosmetic Surfactants and How Do They Work?
Cosmetic surfactants are amphiphilic molecules with a hydrophilic (water-attracting) head and a lipophilic (oil-attracting) tail. This dual structure allows them to reduce surface tension, emulsify oils, and carry both water-soluble and oil-soluble contaminants from skin and hair into the rinse water.
In a cleansing system, surfactants self-assemble into micelles around sebum, makeup residue, and environmental pollutants. Rinsing removes the micelles, along with the material they’ve encapsulated. The efficiency of that process, its net effect on skin barrier function, and the foam profile it produces are all governed by surfactant class, concentration, and the combination of classes in the formula.
Surfactants also appear in non-cleansing applications, serving as emulsifiers, solubilizers, and wetting agents across leave-on and rinse-off formats. The functional ingredients category encompasses this full range. For purposes of this guide, the focus is their role in cleansing systems, where class selection decisions are most consequential.
The Four Ionic Classes of Cosmetic Surfactants
Surfactants used in cosmetics are categorized by the charge of their hydrophilic head group in solution: anionic (negative), cationic (positive), amphoteric (both, depending on pH), or nonionic (none). Each class has a distinct performance profile for foam output, mildness, ionic compatibility, and formulation behavior. Understanding these profiles is the foundation of any surfactant selection decision.

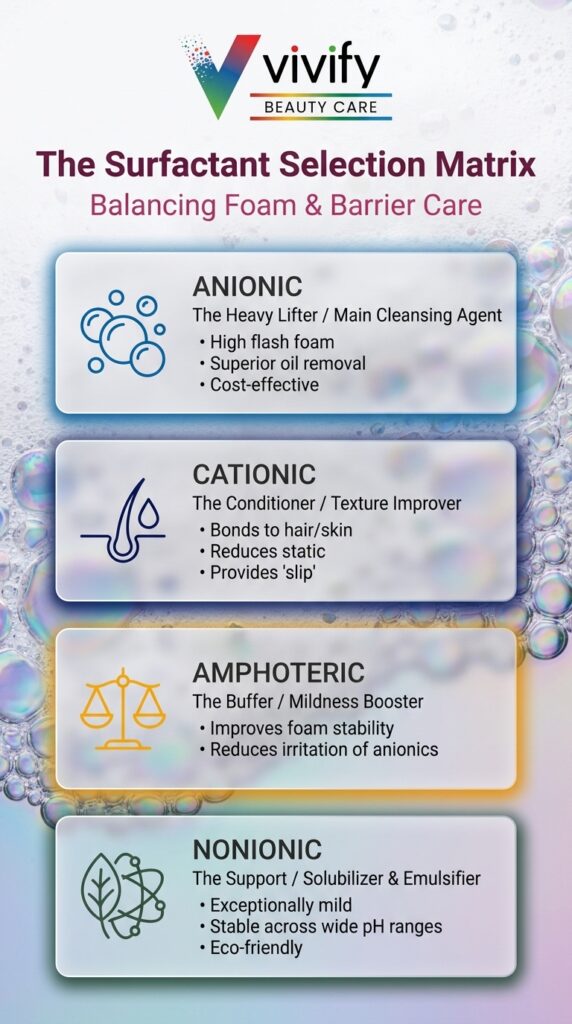
Anionic Surfactants
Anionic surfactants carry a negative charge in solution and are the dominant primary cleansing agents in most rinse-off products. They produce high, rich foam and strong detergency: characteristics that directly drive consumer perception of clean. Sodium lauryl sulfate (SLS) and sodium laureth sulfate (SLES) are the most widely used commercial examples; alkyl sulfates, olefin sulfonates, and ether carboxylates are other common representatives of the class.
The trade-off is skin compatibility. Anionic surfactants are the class most likely to disrupt the skin’s lipid barrier, with potential for transepidermal water loss (TEWL) increase, protein denaturation, and irritation at higher concentrations or under prolonged contact. Their use in sensitive-skin and infant formulations has come under increasing scrutiny, and market demand for alternatives is reflected in the growth of mild-surfactant categories. Formulation operating pH for most anionic systems is 5.0 to 7.0; stability generally decreases below pH 4.5.
Cationic Surfactants
Cationic surfactants carry a positive charge in solution. They adsorb strongly to the negatively charged surface of human skin and hair, which is why they are effective conditioning agents rather than primary cleansers. They do not function well as the main cleansing agent in a rinse-off system and are rarely used in that role.
Quaternary ammonium compounds (quats) are the most common cationic class, used extensively in conditioners, leave-in hair treatments, and antimicrobial products. In a cleansing context, they typically appear at low concentrations as functional additives that improve post-rinse feel, not as primary surfactants. Formulators should also note that cationics are generally incompatible with anionics at equivalent concentrations; precipitation or deactivation can result.
Amphoteric Surfactants
Amphoteric surfactants carry both positive and negative charges, with the net charge determined by the pH of the system. This behavior makes them compatible with all four ionic classes and uniquely useful as co-surfactants in complex formulations.
Their primary role in modern cleansing systems is as a co-surfactant added alongside an anionic primary. Cocamidopropyl betaine (CAPB) is the most commercially significant example. At typical use levels of 3 to 8%, amphoteric co-surfactants reduce the irritation potential of anionic systems, contribute foam stability and creaminess, and add a mild conditioning effect to skin and hair. They are a standard component in facial cleansers, shampoos, and body washes formulated for daily use and broad skin compatibility.
Nonionic Surfactants
Nonionic surfactants carry no charge in solution. They are the mildest class, with minimal irritation potential and excellent compatibility across formulation types. Their cleansing efficacy and foam output are lower than anionics, which limits their usefulness as the sole primary surfactant in most high-foam cleansing applications, but they are highly effective as secondary surfactants, emulsifiers, solubilizers, and foam quality improvers.
Alkyl polyglucosides (APGs) are among the most formulation-relevant nonionic surfactants in current cosmetic use. Derived from glucose and fatty alcohols, they offer a strong sustainability and mildness credential and improve lather quality when combined with anionic primaries. Polysorbates (Tween series) and PEG-based surfactants serve primarily as solubilizers and emulsifiers. In a sulfate-free or low-irritation formula, nonionic surfactants often carry more of the cleansing load than in conventional systems.
Amino Acid Surfactants: Mild Surfactants for Modern Formulation
Amino acid-derived surfactants represent a growing and increasingly commercially important segment within mild surfactants cosmetic formulation. They offer better skin compatibility than conventional anionic surfactants while retaining functional performance across a meaningful pH range, making them well-suited to premium, sensitive-skin, and baby care positioning. Vivify’s mild cleansing agents portfolio includes options within this category.
The three major amino acid surfactant classes used in cosmetics are glutamates, sarcosinates, and isethionates. Each has a distinct performance profile:
| Class | Example INCI | Key Characteristics | Best-Fit Applications |
| Glutamates | Sodium Lauroyl Glutamate | Very low irritation, excellent skin feel, effective pH 5.0–7.0 | Premium facial cleansers, sensitive-skin formulas |
| Sarcosinates | Sodium Lauroyl Sarcosinate | Mild anionic, good foaming at lower concentrations, toothpaste-compatible | Facial cleansers, oral care, sensitive-skin body wash |
| Isethionates | Sodium Cocoyl Isethionate | Moderate foam, creamy lather, very mild, rinse-clean feel | Bar and solid cleansers, baby care, body wash for sensitive skin |
Amino acid surfactants carry a higher raw material cost than SLS/SLES. For brands targeting premium, sensitive-skin, or baby care categories, that cost premium is typically supportable in the market positioning. For mass-market formulations where price efficiency is a constraint, blending a small portion of amino acid surfactant into a mild anionic base can improve the skin compatibility narrative without fully absorbing the cost differential.
How to Choose the Right Surfactant for Your Cleansing Formula
Surfactant selection should be driven by product category, target skin type, foam expectations, pH operating range, and the broader formulation context, including the preservative system and any actives present. There is no universal best surfactant; the right choice is the one that satisfies the performance requirements for a specific formula.
By product category:
- Facial cleansers: Mild anionic or amino acid primary surfactant paired with an amphoteric co-surfactant. Target pH 5.0 to 6.5. Foam should be rich enough to signal efficacy but not aggressive enough to strip the barrier on repeat daily use.
- Shampoos: Higher anionic loading is acceptable in clarifying or weekly-use formulas. Daily-use and scalp-care positioning calls for a mild anionic, amphoteric, and nonionic blend. Pair with appropriate conditioning agents for hair care to manage post-wash feel.
- Body wash: Flexible formula range. Anionic-dominant systems work at scale; APG or amino acid-primary systems support premium or clean-label positioning with a meaningful formulation rationale.
- Baby care and sensitive skin: Nonionic-dominant or amino acid primary systems. Minimize anionic loading. Confirm mildness claims through appropriate in vitro or in vivo testing with appropriate study design for the target claim.
By foam expectation:
- High, rich foam: Anionic primary (SLES at 8 to 15%). Boost foam stability and reduce irritation by adding CAPB at 3 to 8%.
- Moderate, controlled foam: Mild anionic or amino acid primary at lower total surfactant concentrations. APG as secondary surfactant to improve foam quality and mildness.
- Low or no foam: Nonionic-dominant systems. Appropriate for micellar water formats, cleansing oils, or balm-to-milk cleansers where foam is not part of the product concept.
By pH operating range:
Active ingredient compatibility is a critical pH constraint. Formulators working with AHAs, BHAs, or vitamin C derivatives require a low-pH environment; confirm that the selected anionic surfactant is stable across the relevant range before building the formula around it. Some amino acid surfactants (glutamates in particular) offer stability across a wider pH range than conventional anionics and can be useful where pH flexibility is needed.
The Sulfate-Free Formulation Challenge
Removing sulfates from a cleansing system is straightforward in terms of identifying alternative surfactants. The primary technical challenge comes afterward: viscosity. The electrolyte-thickening mechanism that makes NaCl an efficient viscosity builder in SLS/SLES systems loses effectiveness when sulfates are removed. As noted by UL Prospector in their analysis of sulfate-free cleansing: suitable surfactant replacements exist, but building viscosity in a sulfate-free system is the primary formulation hurdle most teams encounter after the initial substitution.
Approaches to address sulfate-free viscosity include:
- Hydroxyethylcellulose (HEC) and other cellulose-based thickeners at appropriate use levels
- Carbomer or acrylate-based polymers neutralized to the target pH
- HPMC or guar derivatives for compatible formula systems
- Electrolyte-alternative thickening agents specifically designed for sulfate-free cleansing systems
For a detailed look at rheology modification strategies compatible with these surfactant systems, see Vivify’s overview of rheology modifiers in cosmetics.
One additional formulation consideration: sulfate-free systems frequently require reassessment of the solubilizer and preservative system. When PEG-based or ethoxylated surfactants are reduced or removed, fragrance and lipophilic active solubilization may be affected. Plan for compatibility testing early.
A note for formulators working across product types: surfactants and emulsifiers overlap in function but differ in application. Surfactants in a rinse-off cleansing system are selected primarily for detergency, foam, and mildness. Emulsifiers in a leave-on system are selected for thermodynamic stability, phase structure, and emulsion type. The FAQ below includes a direct link to Vivify’s emulsifier system guide for formulators who need to navigate both decisions.
Frequently Asked Questions About Cosmetic Surfactants

What are the main types of surfactants used in cosmetics?
There are four primary ionic classes: anionic (negatively charged, highest foam and detergency), cationic (positively charged, primarily conditioning), amphoteric (charge-variable by pH, widely used as co-surfactants), and nonionic (uncharged, mildest irritation profile). A fifth functional category (amino acid-derived surfactants) is technically anionic but distinguished by its mild performance profile and is increasingly specified as a separate class in formulation briefs.
Are surfactants in cosmetics safe for sensitive skin?
Safety for sensitive skin depends on surfactant class, concentration, and formula context. Nonionic and amino acid-derived surfactants have the lowest irritation potential and are the appropriate starting point for sensitive-skin formulations. Anionic surfactants can be used in sensitive-skin products when the concentration is moderated and amphoteric co-surfactants are included to reduce barrier impact. Broad generalizations about surfactant safety, whether positive or negative, are less useful than formulation-specific compatibility and mildness testing.
What is the difference between anionic and nonionic surfactants in cosmetics?
Anionic surfactants carry a negative charge and are the primary cleansing and foaming agents in most rinse-off products; they offer high foam and strong detergency at the cost of higher potential for barrier disruption. Nonionic surfactants carry no charge, are the mildest class, produce significantly less foam, and serve primarily as secondary surfactants, emulsifiers, and solubilizers. In practice, many formulas combine both: an anionic primary for cleansing efficacy and foam, with nonionic secondaries for mildness, compatibility, and texture refinement.
Can surfactants be used as emulsifiers in lotion formulations?
Some surfactants function as emulsifiers in leave-on systems, but the selection criteria are different from those used in cleansing. Emulsifiers are chosen for their ability to form and stabilize an emulsion structure over the product’s shelf life; foaming, detergency, and skin-feel-after-rinse are not relevant parameters. Certain nonionic surfactants (polysorbates, sorbitan esters) are used specifically as emulsifiers. For a full treatment of emulsifier selection, see Vivify’s emulsifier system guide.
What is the role of surfactants in skin irritation and acne-prone formulations?
Surfactants do not treat or prevent acne, but surfactant selection directly affects skin barrier integrity and irritation risk, both of which are relevant considerations for acne-prone formulations. High-concentration anionic surfactants can increase transepidermal water loss (TEWL) and disrupt the acid mantle, creating conditions that can worsen irritation in sensitized or acne-prone skin. Formulating with milder alternatives, such as amino acid-derived surfactants, amphoteric co-surfactants, or nonionic-dominant blends, reduces barrier disruption. Maintaining a cleansing formula pH of 5.0 to 5.5 further supports acid mantle preservation, and that combination of surfactant class and pH management represents the primary lever available at the formulation stage.
Explore Vivify’s Surfactant Portfolio
Whether you’re formulating a sulfate-free body wash, a mild facial cleanser, or a daily shampoo with a sensitive-scalp claim, surfactant selection is where the formula’s performance story starts. Vivify Beauty Care’s surfactants and cleansing agents portfolio includes options across ionic classes with filtering by certification status, physical form, and product type suitability.
Need help evaluating surfactant options for a specific application, or troubleshooting a sulfate-free viscosity challenge? Vivify’s formulation support team works directly with R&D and product development teams across formula types and scales. Reach out to a Vivify technical specialist to get started.
Disclaimer
The information provided in this blog is intended for general informational purposes only and is furnished without warranty, expressed or implied. The content reflects insights and information accurate to the best knowledge of Vivify Beauty Care at the time of publication.
This blog content should be used as a general guide and does not constitute a substitute for direct professional advice or product-specific consultation. Vivify Beauty Care does not validate any claims made within the blog, and customers bear the ultimate responsibility for ensuring their product applications and associated claims are compliant with all applicable laws and regulations. For specific inquiries or tailored recommendations regarding our product specifications and service offerings, please contact our sales professionals.



