Rheology modifiers in cosmetics do far more than increase viscosity. They control emulsion stability, define sensory properties during application, suspend active particles, and influence long-term shelf performance. Yet many formulation teams select thickeners based on familiarity or supplier availability rather than matching the modifier to the system’s structural requirements.
The result is predictable: separation during accelerated stability, syneresis on the shelf, or a stringy texture that consumers reject. These failures rarely stem from using “not enough thickener.” They stem from choosing the wrong thickening mechanism for the formulation architecture at hand.
This guide takes a systems-level approach to rheology modifier selection. Rather than listing ingredients alphabetically, it organizes choices by thickening mechanism, formulation system, and observed failure mode so you can diagnose problems, select the right structural tool, and optimize both stability and sensory outcomes.
Key Takeaways
- Rheology modifiers work through three primary mechanisms: chain entanglement, cross-linked network swelling, and associative thickening. Selecting the right mechanism for your system matters more than selecting a specific ingredient.
- Carbomers offer superior gel clarity and thickening efficiency at 0.1–0.5% use levels but lose performance dramatically in high-electrolyte systems (above approximately 1–2% NaCl equivalent).1
- Xanthan gum provides broad pH stability (pH 2–12), strong pseudoplastic flow, and excellent electrolyte tolerance, making it the most versatile natural-origin rheology modifier for cosmetic emulsions.2
- Most common rheology failures, including creaming, syneresis, and viscosity drift, trace back to a mismatch between the thickening mechanism and the formulation’s ionic strength, pH, or oil-phase ratio.
- Combining modifiers from different mechanistic classes (for example, a carbomer with xanthan gum) often delivers better stability and sensory balance than increasing the concentration of a single thickener.3
What Are Rheology Modifiers and Why Do They Matter Beyond Viscosity?
Rheology modifiers are ingredients that alter the flow and deformation behavior of a formulation. In cosmetic applications, they serve multiple functions simultaneously:
- Viscosity build. Increasing the apparent thickness of the continuous phase to achieve the desired product body.
- Emulsion stabilization. Developing yield stress in the continuous phase to prevent droplet creaming, coalescence, and phase separation.4
- Particle suspension. Holding insoluble materials (pigments, exfoliants, mineral sunscreen actives) uniformly distributed throughout the product.
- Sensory shaping. Defining spreadability, skin feel, pick-up, and the perceived richness or lightness of the product on application.
The term “thickener” is commonly used interchangeably with “rheology modifier,” but this simplification can lead to poor formulation decisions. A modifier that increases apparent viscosity at low shear may do nothing to prevent creaming at rest if it fails to develop adequate yield stress. Similarly, a modifier that builds impressive viscosity in a beaker may impart an unacceptable stringy or tacky skin feel.
Effective selection requires understanding not just how much viscosity a modifier provides, but how it builds structure and where that structure performs across different shear regimes. A thickener that stabilizes an emulsion cream may perform poorly in a surfactant-based cleanser, and a modifier that builds clarity in a serum gel may fail entirely when electrolytes are introduced by active ingredients.
Thickening Mechanisms: How Different Rheology Modifiers Build Structure

Understanding the mechanism behind viscosity build is the foundation of effective rheology modifier selection. Cosmetic thickeners generally operate through one of three structural mechanisms, each with distinct strengths, limitations, and formulation implications.5
Chain Entanglement
Linear or branched polymers dissolved in the continuous phase increase viscosity through physical entanglement of polymer chains. As concentration rises, chains overlap and intertwine, restricting the flow of the surrounding solvent. Common chain-entanglement modifiers include:
- Hydroxyethyl cellulose (HEC): 0.5–2.0% use level; moderate viscosity build; good surfactant compatibility.
- Hydroxypropyl methylcellulose (HPMC): 0.5–1.5% use level; clear gels; stable in alcohol-based systems.
- Hydroxypropyl guar gum: 0.2–1.0% use level; conditioning properties; suited to hair care emulsions and cleansers.
These modifiers produce shear-thinning behavior: viscosity decreases under applied shear (during rubbing or spreading) and recovers at rest. Their limitations include potential for “slimy” or “stringy” sensory textures at higher concentrations and moderate thickening efficiency compared with cross-linked alternatives.
Cross-Linked Network Swelling
Cross-linked polymers, most notably carbomers (cross-linked polyacrylic acid), swell in aqueous media upon neutralization, forming a three-dimensional microgel network that dramatically increases viscosity. A 0.2–0.5% carbomer dispersion, once neutralized to pH 5–8, can produce viscosities exceeding 40,000 cP.1
Key characteristics of cross-linked network thickeners:
- High efficiency. Small additions yield large viscosity increases, making carbomers cost-effective at 0.1–0.5% use levels.
- Clear gels. Carbomers produce transparent to near-transparent gels, ideal for serum and gel formats.
- Short-flow rheology. Gels break cleanly on application rather than forming strings, a texture most consumers prefer in leave-on products.
- Electrolyte vulnerability. Divalent cations (Ca²⁺, Mg²⁺) and monovalent salts above approximately 1–2% NaCl screen the charge repulsion that drives network swelling, collapsing viscosity and potentially causing precipitation.6
This electrolyte sensitivity makes carbomers a poor standalone choice in formulations containing mineral sunscreens, AHA salts, or high-electrolyte botanical extracts.
Associative Thickening
Associative thickeners contain hydrophobic groups distributed along a hydrophilic polymer backbone. In aqueous systems, the hydrophobic regions aggregate in a manner analogous to surfactant micelle formation, creating a physical network that increases viscosity.5 The most commonly used associative rheology modifiers in cosmetics include:
- Acrylates/C10-30 alkyl acrylate crosspolymers: Widely used; better electrolyte tolerance than conventional carbomers; interact synergistically with surfactant systems.
- Hydrophobically modified ethoxylated urethanes (HEUR): Nonionic associative thickeners; salt and pH stable; non-tacky skin feel.
- Hydrophobically modified alkali-swellable emulsions (HASE): Combine acid-swellable and associative mechanisms for versatile performance.
Associative modifiers produce pleasant sensory profiles with good spreadability. However, their performance is more system-dependent: changes that alter the hydrophobic environment (adding oils, adjusting surfactant ratios) can unpredictably increase or decrease viscosity. Bench-scale testing against your specific formula base is essential.
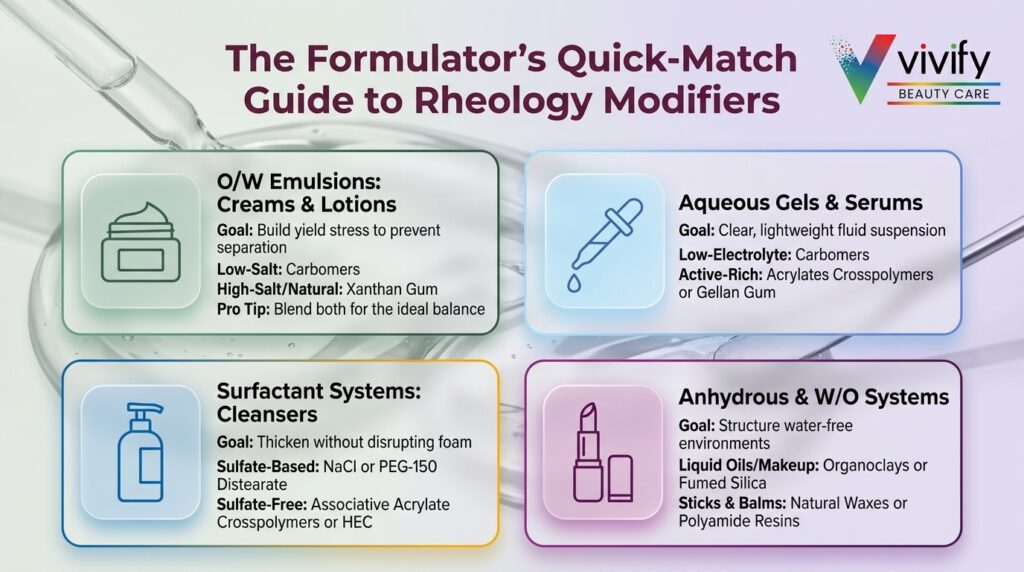
Rheology Modifier Selection by Formulation System

No single rheology modifier performs optimally across all cosmetic formats. The continuous phase, oil-phase ratio, surfactant load, pH, and ionic strength all influence which thickening mechanism will deliver the best stability and sensory result.
O/W Emulsions: Creams, Lotions, and Milks
Oil-in-water emulsions are the most common cosmetic format and typically require rheology modifiers that build yield stress in the continuous aqueous phase to prevent droplet creaming and coalescence. Recommended approaches include:
- Carbomers (0.1–0.4%): High efficiency, clear contribution, short-flow texture. Best suited for low-electrolyte emulsions.
- Xanthan gum (0.2–0.5%): Robust electrolyte tolerance, strong pseudoplastic flow, broad pH stability. Preferred where natural-origin certification or electrolyte compatibility is required.2
- Carbomer/xanthan blends: Combine carbomer’s gel clarity and efficiency with xanthan’s electrolyte resilience. This combination also reduces yield-stress failure during thermal cycling, a common issue with carbomer-only systems.3
The emulsifier system also contributes to overall rheology architecture. Self-emulsifying waxes and high-HLB emulsifiers create their own lamellar or crystalline structures that supplement the modifier’s contribution. Factor in the emulsifier’s structural role when determining thickener concentrations to avoid over-thickening.
Aqueous Gels and Serums
Clear aqueous gels demand modifiers that produce transparent or translucent systems. For serum and essence formats, use levels are typically lower (0.1–0.3%) to achieve a light, fluid viscosity that still prevents sedimentation of active particles. Modifier selection depends on the formulation’s electrolyte load:
- Low-electrolyte gels: Carbomers remain the benchmark for clarity and short-flow texture. Pre-neutralized sodium carbomer grades offer comparable clarity with simpler cold-process manufacturing.
- Moderate-electrolyte gels (niacinamide, ascorbic acid salts, AHA salts): Acrylates/C10-30 alkyl acrylate crosspolymers or high-acyl gellan gum (0.3–0.5%) provide more stable viscosity build under ionic stress.7
- High-electrolyte gels: Xanthan gum or cellulose derivatives (HEC, HPMC) tolerate high salt loads where carbomers fail entirely.
Surfactant-Based Systems: Cleansers, Shampoos, and Body Washes
Surfactant systems require rheology modifiers that thicken without disrupting foam structure or defoaming the product. NaCl is the traditional viscosity builder for anionic surfactant systems (typically 1–3%), but it has a narrow effective range and can cause viscosity drop at higher concentrations due to micellar transition. Polymer-based alternatives include:
- Associative acrylate crosspolymers: Interact synergistically with surfactant micelles; build viscosity without electrolyte dependence.
- HEC (0.5–1.5%): Reliable viscosity build independent of electrolyte concentration; widely compatible with surfactant bases.
- PEG-150 distearate: Effective co-thickener in sulfate-based systems; adds pearlescence.
For sulfate-free systems that lack the easy NaCl thickening response of sulfate-based surfactants, polymer-based rheology modifiers become essential for achieving the viscosity consumers expect.
Anhydrous and W/O Systems
Water-soluble polymers are ineffective in anhydrous systems. Oil-phase rheology modification relies on a different set of structurants:
- Organoclays (modified hectorite, bentonite): 1–4% use level; build thixotropic structure; suspend pigments in liquid lipsticks and foundations.
- Fumed silica: 0.5–3% use level; thixotropic; effective across a wide range of oil polarities.
- Wax structurants (beeswax, candelilla, carnauba): 5–20% of formula; provide hardness and shape retention in sticks and balms.
- Polyamide resins: 2–8% use level; gel non-polar oils; create elegant, non-waxy textures in lip oils and treatment oils.

Troubleshooting Common Rheology Failures
Observed stability or sensory defects in a finished product frequently point to a specific rheological root cause. The following diagnostic framework connects common failure modes to their most likely structural origins and corrective actions.
Phase Separation and Creaming
Creaming occurs when the yield stress of the continuous phase is insufficient to counteract the buoyancy-driven rise of oil droplets. Before adding more thickener, confirm the root cause is rheological rather than emulsifier-related (if the emulsion is coalescing, increasing viscosity will only delay failure). When the issue is yield-stress driven, corrective options include:
- Adding or increasing a modifier with strong yield-stress character: xanthan gum (even 0.1–0.2% addition can significantly boost yield stress),2 a carbomer at adequate neutralization, or microcrystalline cellulose as a co-stabilizer.
- Reducing droplet size through improved homogenization, which decreases the buoyancy force acting on each droplet.
- Avoiding sole reliance on chain-entanglement thickeners like HEC for anti-creaming performance; they build viscosity at higher shear but often develop inadequate yield stress at rest.
Syneresis (Water Release)
Syneresis, the expulsion of water from a gel or emulsion surface, typically indicates that the polymer network is contracting or that the system lacks adequate water-binding capacity. Common causes and corrective approaches:
- Over-neutralization of carbomer gels (pH above 8). Verify pH is within the modifier’s optimal functional range (pH 5–8 for most carbomers).
- Electrolyte contamination. Reduce ionic strength or switch to an electrolyte-tolerant modifier.
- Insufficient water binding. Increase humectant load (glycerin, propanediol, butylene glycol) to compete for water binding.
- Freeze-thaw damage to polysaccharide networks. Add a secondary polymer with complementary water-holding properties or reformulate with a freeze-thaw-stable modifier.
Stringy, Slimy, or Tacky Texture
Stringiness is characteristic of high-molecular-weight linear polymers at moderate to high concentrations. Xanthan gum above approximately 0.5% is a frequent source.8 HEC and certain guar derivatives can produce similar textures. To correct:
- Reduce the concentration of the offending polymer and supplement with a cross-linked modifier (carbomer or acrylates crosspolymer) that provides “short flow” rather than “long flow” rheology.
- Blend with a sensory modifier such as microcrystalline cellulose to interrupt the stringy texture while maintaining stability.
- Consider switching to a pre-blended rheology system (e.g., acacia/xanthan gum blends) engineered to deliver xanthan’s stability advantages with improved skin feel.
Viscosity Loss Over Time or During Processing
Gradual viscosity decline during shelf storage or sudden loss during manufacturing can have several root causes:
- Microbial degradation. Natural gums are susceptible to enzymatic breakdown without adequate preservation. Monitor microbial counts alongside viscosity in stability studies.
- Electrolyte migration. Encapsulated actives, time-release systems, or pH-buffering salts can slowly release ions that suppress carbomer performance over weeks to months.
- Excessive processing shear. High-shear homogenization can permanently degrade cross-linked polymer networks. Add the rheology modifier after the high-shear step when process flow allows, or select shear-resistant grades.2
- pH drift. Monitor pH at regular intervals during accelerated stability testing. Even small pH shifts can affect carbomer performance.
Electrolyte Compatibility: The Most Overlooked Selection Criterion
Electrolyte compatibility is arguably the single most important and most frequently underestimated factor in cosmetic rheology modifier selection. Many formulation failures attributed to “instability” are actually electrolyte-induced rheology collapse.6
The following table summarizes electrolyte tolerance across major rheology modifier categories. Use it as a first-pass screening tool when your formulation contains salts, mineral sunscreen actives (ZnO, TiO₂), or ionic active ingredients.
| Modifier Class | Electrolyte Tolerance | pH Range | Key Limitation |
| Carbomers | Low | 5–8 | Viscosity collapse above ~1–2% NaCl equivalent; precipitates with divalent cations |
| Xanthan gum | High | 2–12 | Tacky texture above ~0.5%; incompatible with some cationic ingredients |
| HEC / HPMC | Moderate | 3–11 | Moderate efficiency; can produce stringy texture at higher concentrations |
| Acrylates/C10-30 alkyl acrylate crosspolymer | Moderate–High | 4–10 | System-dependent; viscosity varies with oil phase and surfactant ratios |
| High-acyl gellan gum | Moderate | 4–8 | Soft gels only; limited high-viscosity build at low concentrations |
| Organoclays (bentonite, hectorite) | N/A (oil phase) | N/A | Oil-phase only; requires activation with polar solvents; can feel heavy |
When working with electrolyte-rich formulations, default to xanthan gum, associative crosspolymers, or cellulose derivatives rather than conventional carbomers. If carbomer clarity and texture are essential, test an acrylates/C10-30 alkyl acrylate crosspolymer, which retains associative thickening in moderate-electrolyte environments where conventional carbomers fail.
Sensory Considerations: Matching Rheology to Consumer Expectations
Rheology and sensory perception are directly linked. Oscillatory rheology measurements (storage modulus G′, loss modulus G″, and loss tangent tan δ) correlate with consumer-perceived attributes such as firmness, spreadability, and richness.4 Use these relationships to target specific sensory outcomes:
- Light, serum-like feel: Low-concentration carbomer or acrylate crosspolymer systems (0.1–0.25%) that yield low viscosity with clean break on application.
- Rich cream or mask texture: Higher concentrations of xanthan gum or carbomer blends (0.3–0.5%) that produce elevated yield stress and a cushioned feel.
- Gel-cream hybrid: Moderate structure with strong shear-thinning. Combine a carbomer (for short flow) with a low level of xanthan (for yield stress) to feel substantial yet glide without drag.
Blending modifiers from different classes is one of the most effective strategies for sensory optimization. A small addition of an associative crosspolymer to a xanthan-based emulsion can reduce the tackiness that xanthan imparts while maintaining the stability advantages of its pseudoplastic flow profile. Vivify’s portfolio of texture and sensory ingredients and performance enhancement solutions can complement rheology modifier selection to achieve targeted skin-feel outcomes.

Frequently Asked Questions About Rheology Modifiers in Cosmetics
Can I replace carbomer with xanthan gum in an existing formulation?
Not as a direct 1:1 substitution. Carbomers and xanthan gum operate through different thickening mechanisms and produce different rheological profiles. Carbomers create short-flow, clear gels; xanthan gum produces pseudoplastic, slightly tacky systems. A switch typically requires adjusting concentration, potentially blending with a secondary modifier for sensory balance, and re-running stability testing. Xanthan gum also does not require neutralization, so the formulation’s pH buffering system may need revision.
Why does my gel lose viscosity when I add vitamin C or niacinamide?
Both ascorbic acid (and its salts) and niacinamide introduce electrolytes into the aqueous phase. If your gel relies on a carbomer for viscosity, the ionic species screen the polymer’s carboxylate charges, reducing chain expansion and collapsing the gel network.6 Switch to an electrolyte-tolerant modifier (acrylates/C10-30 alkyl acrylate crosspolymer, xanthan gum, or gellan gum) or use a carbomer/associative thickener blend that maintains viscosity under ionic stress.
What is the difference between short-flow and long-flow rheology?
Short-flow rheology describes a gel or cream that breaks cleanly when dispensed or scooped, holds its shape briefly, then flows in a controlled, non-stringy manner. Carbomers are the classic example. Long-flow rheology describes a system that forms strings or threads when stretched, characteristic of high-molecular-weight linear polymers like xanthan gum, HEC, and guar gum at higher concentrations. Consumer preference generally favors short flow for leave-on products.
How do I thicken a surfactant system without using salt?
Polymer-based thickeners offer salt-free viscosity building for surfactant and cleansing systems. Associative crosspolymers work synergistically with anionic surfactant micelles. HEC (0.5–1.5%) provides reliable viscosity build independent of electrolyte concentration. PEG-150 distearate is another effective option, particularly in sulfate-based systems.
Do natural-origin thickeners perform as well as synthetic ones?
Performance depends on the application. Xanthan gum matches or exceeds synthetic polymers in electrolyte tolerance, pH stability, and suspension capability. However, carbomers still offer superior gel clarity, thickening efficiency at low concentrations, and short-flow texture that natural alternatives have not fully replicated. The best approach for many formulations is a blend that leverages the strengths of both natural and synthetic modifiers while meeting the product’s regulatory and marketing requirements.
Build More Stable, Sensory-Optimized Formulations
Selecting the right rheology modifier is a formulation decision that affects every aspect of product performance, from shelf stability to consumer satisfaction. Whether you are troubleshooting a viscosity failure, reformulating for electrolyte compatibility, or developing a new texture profile, Vivify’s technical team can help you evaluate options across our functional ingredients portfolio. Connect with a Vivify formulation specialist to request samples or discuss your next project.
References
1. Islam, M. T., Rodriguez-Hornedo, N., Ciotti, S., & Ackermann, C. (2004). Rheological characterization of topical carbomer gels neutralized to different pH. Pharmaceutical Research, 21(7), 1192–1199. https://doi.org/10.1023/B:PHAM.0000033006.11619.07
2. García-Ochoa, F., Santos, V. E., Casas, J. A., & Gómez, E. (2000). Xanthan gum: Production, recovery, and properties. Biotechnology Advances, 18(7), 549–579. https://doi.org/10.1016/S0734-9750(00)00050-1
3. Piao, L., Park, H., & Jo, C. H. (2021). Agarose stearate–carbomer940 as stabilizer and rheology modifier for surfactant-free cosmetic formulations. Polymers, 13(13), 2074. https://doi.org/10.3390/polym13132074
4. Gilbert, L., Picard, C., Savary, G., & Grisel, M. (2013). Rheological and textural characterization of cosmetic emulsions containing natural and synthetic polymers: Relationships between both data. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 421, 150–163. https://doi.org/10.1016/j.colsurfa.2013.01.003
5. Ferraris, C., Ricci, A., Provenzale, M., et al. (2025). On the key role of polymeric rheology modifiers in emulsion-based cosmetics. Cosmetics, 12(2), 76. https://doi.org/10.3390/cosmetics12020076
6. Fresno Contreras, M. J., Ramírez Dieguez, A., & Jiménez Soriano, M. M. (2001). Viscosity and temperature relationship in ethanol/water mixtures of carbomers. Il Farmaco, 56(5–7), 443–445. https://doi.org/10.1016/S0014-827X(01)01068-X
7. Morris, E. R., Nishinari, K., & Rinaudo, M. (2012). Gelation of gellan: A review. Food Hydrocolloids, 28(2), 373–411. https://doi.org/10.1016/j.foodhyd.2012.01.004
8. Sworn, G. (2009). Xanthan gum. In G. O. Phillips & P. A. Williams (Eds.), Handbook of hydrocolloids (2nd ed., pp. 186–203). Woodhead Publishing. https://doi.org/10.1533/9781845695873.186
Disclaimer
The information provided in this blog is intended for general informational purposes only and is furnished without warranty, expressed or implied. The content reflects insights and information accurate to the best knowledge of Vivify Beauty Care at the time of publication.
This blog content should be used as a general guide and does not constitute a substitute for direct professional advice or product-specific consultation. Vivify Beauty Care does not validate any claims made within the blog, and customers bear the ultimate responsibility for ensuring their product applications and associated claims are compliant with all applicable laws and regulations. For specific inquiries or tailored recommendations regarding our product specifications and service offerings, please contact our sales professionals.

