For any product positioned as water resistant or sweat resistant, the path from bench to market requires three things to align: formulation architecture that genuinely performs, a validated test method that generates defensible data, and label language that stays within regulatory boundaries in the U.S. and EU. Water resistance claims cosmetics brands need to substantiate begin with the formula, not the marketing brief.
Most available guidance covers only one piece of that chain. Regulatory guides explain what you can and cannot say. Testing lab pages describe their protocols. Ingredient pages detail film former performance. This post connects all three: ingredient and architecture choices that drive water and sweat resistance, the test methods that validate them, and the specific claim language your label can carry.
Key Takeaways
- “Waterproof” and “sweatproof” are explicitly prohibited label terms for OTC sunscreens under the FDA’s 2011 final rule. The only permitted water resistance claims for sunscreens are “Water Resistant (40 minutes)” or “Water Resistant (80 minutes).”
- For non-sunscreen cosmetics (foundations, mascaras, lip products), “waterproof” is not explicitly banned in the U.S., but it requires substantiation with reliable test data under FTC guidelines and FDA cosmetic labeling regulations.
- EU Cosmetics Regulation (EC 1223/2009) requires all claims, including water resistance language, to be truthful, evidenced, honest, fair, and informed. Unsubstantiated claims create regulatory exposure in both U.S. and EU markets.
- Film formers are the primary formulation lever for water and sweat resistance. Key categories include silicone resins (trimethylsiloxysilicate), acrylate/styrene copolymers, polyurethane dispersions, and VP/VA copolymers, each with different performance profiles and formulation trade-offs.
- Three test methods apply by product category: ISO 18861 for sunscreen water resistance, validated in vitro perspiration protocols for non-sunscreen sweat resistance, and the FDA 21 CFR 201.327 immersion protocol for OTC sunscreen label claims in the U.S.
- Claims substantiation documentation is a legal requirement. MoCRA (U.S.) and the EU Product Information File (PIF) require brands to maintain test data, protocols, and results on file. Gaps in documentation are a leading cause of regulatory inquiry.
Why Water and Sweat Resistance Claims Require More Than Marketing Language
Water and sweat resistance claims are among the most closely scrutinized statements in cosmetic and OTC drug labeling. A product positioned as water resistant is making a verifiable performance promise, and both U.S. and EU regulators treat that promise as a claim requiring documented support.
U.S. Framework: FDA and FTC Oversight
In the U.S., manufacturers are legally responsible for ensuring all product labeling is truthful and not misleading. The FDA’s guidance on cosmetics labeling claims affirms that performance claims must be supportable, and the FTC independently requires that objective claims be substantiated by competent and reliable scientific evidence. For OTC sunscreen products, the FDA has codified prohibited terms and required test methods at the product level, raising the bar above what applies to standard cosmetics.
EU Framework: EC 1223/2009 Common Criteria
In the EU, Article 20 of EC 1223/2009 prohibits claims that attribute characteristics a product does not possess. The associated Common Criteria require that claims be truthful, evidenced, honest, fair, and informed. Aspirational water resistance language without documented test support creates direct regulatory exposure in all EU markets.
Water Resistant vs. Waterproof: What You Can and Cannot Say
The distinction between “water resistant” and “waterproof” is legally significant, and the rules differ meaningfully by product category.
OTC Sunscreens: Prohibited Terms and Permitted Claims
The FDA’s Q&A on OTC sunscreen requirements states: “Manufacturers cannot label sunscreens as ‘waterproof’ or ‘sweatproof,’ or identify their products as ‘sunblocks’… unless FDA approves such claims.” This prohibition applies specifically to OTC sunscreen products. The only permitted water resistance designations are:
- Water Resistant (40 minutes)
- Water Resistant (80 minutes)
These designations map directly to performance outcomes under the FDA’s water resistance testing protocol at 21 CFR 201.327. A product that does not pass the required water immersion test may not carry any water resistance claim on its label.
Non-Sunscreen Cosmetics: A Different Regulatory Landscape
For non-sunscreen cosmetics (foundations, mascaras, lip products), U.S. law does not explicitly prohibit “waterproof.” The claim still requires FTC substantiation. Any objective performance claim must be supported by competent and reliable test data, and claims that exceed what testing demonstrates create compliance risk. In the EU, the Common Criteria framework applies equally across all cosmetic categories: “waterproof” language on any product must be backed by reproducible testing aligned with normal conditions of use.
Building the Formula: Ingredient Strategies for Water and Sweat Resistance
Film former selection and emulsion type are the two formulation variables with the greatest influence on water and sweat resistance test performance. Getting both right before stability testing begins avoids costly reformulation cycles.
Film Formers: The Primary Performance Lever
Film formers create water and sweat resistance by forming a continuous, hydrophobic barrier on the skin, lip, or lash surface that resists displacement by water. The main categories used in water-resistant cosmetics include:
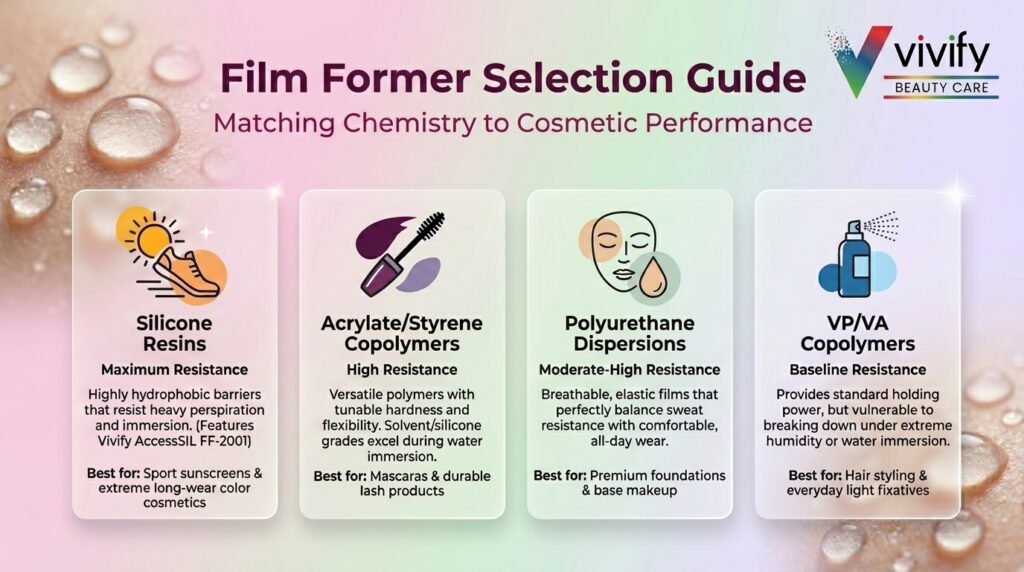

- Silicone resins (trimethylsiloxysilicate, MQ resins): Highly hydrophobic film formers with strong resistance to water and perspiration. Well-suited for sport sunscreens and long-wear color cosmetics. Vivify’s AccessSIL FF-2001 forms a hydrophobic barrier resistant to water and sweat across those application categories.
- Acrylate/styrene copolymers: Versatile film formers with tunable hardness and flexibility. Common in mascara and lash products. Solvent-borne and silicone-based grades outperform water-dispersible versions in immersion conditions.
- Polyurethane dispersions: Flexible, breathable films with good adhesion and sweat resistance, favored in foundations where wear comfort and longevity are both priorities.
- VP/VA copolymers: Common in styling and fixative applications. Provide moderate water resistance but are less effective than silicone resins under high-humidity or immersion conditions.
Vivify’s portfolio of skin film formers and broader film formers and fixatives spans these categories with options designed for color cosmetic and sun care applications.
Emulsion Architecture and the Rheology Trade-Off
Water-in-silicone (W/Si) emulsions provide inherently greater water and sweat resistance than oil-in-water (O/W) systems because the continuous silicone phase forms a hydrophobic matrix around the dispersed water droplets. Emulsifier selection directly determines which architecture is achievable. See the guide to choosing the right emulsifier system for detailed formulation context.
Rheology modification and water resistance are frequently in tension. Thickeners and structuring agents that improve emulsion stability can interfere with the coherent film a film former needs to establish on the skin surface. An industry analysis in Cosmetics & Toiletries (January 2020) documented this trade-off across product formats and testing standards. Evaluating stability and water resistance in parallel rather than sequentially avoids reformulation cycles late in development.
Selecting the Right Test Method for Water Resistance Claims
No single test method applies across all product categories and claim types. Method selection is determined by product type, target market, and the specific claim language you plan to support. Applying the wrong method generates data that does not substantiate your intended claim.
ISO 18861: Sunscreen Water Resistance
ISO 18861 is the primary standardized method for measuring water resistance in sunscreen products. The test measures SPF before and after water immersion under controlled conditions; a product must generally retain at least 50% of its pre-immersion SPF to substantiate a claim. ISO 18861 is widely accepted in the EU and international markets. Active work from the Cosmetics Europe Sun Protection Methods Task Force is developing validated in vitro alternatives: a ring-test validation study published in the International Journal of Cosmetic Science demonstrated that a new in vitro approach can reliably determine sunscreen water resistance across multiple test centers.1
Sweat Resistance Testing for Non-Sunscreen Cosmetics: Industry Methods
No single mandatory ISO standard governs sweat resistance testing for non-sunscreen cosmetics. Unlike sunscreen water resistance, which is governed by ISO 16217 and ISO 18861, sweat resistance testing for foundations, color cosmetics, and other non-drug products relies on validated in vitro protocols developed or adopted by the testing laboratory. The general methodology is consistent across labs: a cosmetic sample is applied to a substrate that mimics skin, subjected to artificial perspiration under controlled temperature and humidity, and evaluated for color change, transfer, and visible alteration. Results are reproducible when the protocol is documented and the lab operates under ISO 17025 or equivalent quality systems. Both the U.S. and EU accept this category of validated laboratory data as adequate substantiation for sweat resistance claims on non-drug cosmetics.
FDA 21 CFR 201.327: The U.S. OTC Sunscreen Protocol
For OTC sunscreen products sold in the U.S., the FDA’s 21 CFR 201.327 protocol is the required in vivo procedure. Participants enter a whirlpool bath for timed intervals: two 20-minute periods for the 40-minute claim, four 20-minute periods for the 80-minute claim. SPF is measured before and after immersion, and the post-immersion value determines the permitted label claim. This protocol differs procedurally from ISO 18861 and is not interchangeable for U.S. label purposes.
From Test Data to Label Claims: Translating Results into Compliant Language

Passing a water resistance test does not automatically determine what your label can say. The mapping from test outcome to permitted claim language is specific and, for OTC sunscreens, non-negotiable.
OTC Sunscreens: The Label Language Is Fixed
For U.S. OTC sunscreens, only the exact phrases “Water Resistant (40 minutes)” and “Water Resistant (80 minutes)” are compliant. Any variation, including paraphrased or abbreviated versions, is not permitted. If a product does not achieve SPF retention at either interval, no water resistance claim may appear on the label.
Non-Sunscreen Cosmetics: Flexible but Bounded
For non-sunscreen cosmetics, permitted claim language is more flexible but still bounded by the strength of your test evidence. “Waterproof” implies complete resistance and requires data matched to actual conditions of use. Claims that overstate performance relative to test results create FTC and FDA exposure in the U.S. and EU Common Criteria exposure in European markets.
Ingredient selection affects claim scope as well. Products formulated with performance-enhancing functional ingredients and color cosmetic-specific functional ingredients should be tested against methods appropriate to the product format, not a generic protocol that may not reflect actual use conditions.
Claims Substantiation Documentation: What to Keep on File
Claims substantiation is not complete when the test is finished. Both the U.S. and EU require brands to maintain specific records and to produce them upon request.
U.S. Requirements Under MoCRA
The Modernization of Cosmetics Regulation Act of 2022 (MoCRA) reinforces the requirement that product labeling be truthful and not misleading. The FDA’s product testing guidance confirms that manufacturers bear responsibility for substantiating their claims. Brands should maintain at minimum:
- The test protocol and methodology used
- The testing laboratory’s identity and accreditation status
- The raw data and final test report
- The formulation batch that was tested
- A clear mapping of the test result to the specific label claim language
EU Requirements: The Product Information File
All cosmetic products must have a Product Information File (PIF) in place before market placement under EC 1223/2009. The PIF must include the test method, protocol, data, and a rationale for how the results support the specific claim language. The Responsible Person bears legal accountability for PIF completeness.
For products targeting both markets, a harmonized substantiation package covering FDA 21 CFR 201.327 data where applicable alongside ISO test results and EU PIF-compatible documentation reduces redundant testing and simplifies regulatory review.
Frequently Asked Questions About Water Resistance Claims in Cosmetics
What is the difference between water resistant and waterproof cosmetics?
“Water resistant” indicates tested performance under defined water exposure conditions and is the only permissible terminology for OTC sunscreens under FDA’s 2011 final rule, with the claim duration (40 or 80 minutes) required on the label. “Waterproof” implies complete resistance to water penetration. For OTC sunscreens, it is explicitly prohibited in the U.S. For non-sunscreen cosmetics, “waterproof” may be used if substantiated by appropriate test data, though it carries a higher substantiation burden given the strength of the implied performance claim.
What does ISO 18861 actually test?
ISO 18861 measures SPF retention in sunscreen products after controlled water immersion. The test establishes a pre-immersion SPF baseline, subjects the product to standardized water exposure, and measures post-immersion SPF. A product must generally retain at least 50% of its pre-immersion value to substantiate a water resistance claim under this standard. ISO 18861 is widely used internationally but is procedurally distinct from the FDA’s in vivo 21 CFR 201.327 protocol, which is specifically required for U.S. OTC sunscreen label claims.
Can makeup products still be called “waterproof”?
Yes, with substantiation. For non-sunscreen cosmetics, “waterproof” is not prohibited under U.S. cosmetic law, but any objective performance claim requires competent and reliable test data under FTC guidelines. In the EU, the claim must comply with the Common Criteria of EC 1223/2009: it must be truthful and backed by reproducible testing results appropriate to the product’s intended conditions of use. A validated in vitro perspiration protocol conducted by an ISO 17025-accredited laboratory is appropriate substantiation in both markets.
How do film formers create water resistance in cosmetic formulations?
Film formers deposit a continuous polymer matrix on the skin, lip, or lash surface that physically resists displacement by water. Effectiveness depends on film cohesion, flexibility, and hydrophobicity. Silicone resins (trimethylsiloxysilicate) produce highly hydrophobic films with strong resistance to water and perspiration. Acrylate and polyurethane systems offer greater flexibility but vary in resistance by chemistry and grade. W/Si emulsion architecture improves baseline resistance beyond what film former selection alone can achieve.
What testing is required for sweat-resistant makeup claims?
There is no single mandatory ISO standard for sweat resistance testing in non-sunscreen cosmetics. Unlike sunscreen water resistance, which is governed by ISO 16217 and ISO 18861, non-sunscreen sweat resistance relies on validated in vitro perspiration protocols: a cosmetic sample is applied to a skin-mimicking substrate, subjected to artificial perspiration under controlled temperature and humidity, and evaluated for color change and visible alteration. In the U.S., the FDA does not mandate a specific protocol for non-sunscreen sweat resistance claims; a rigorously documented, validated method conducted by an ISO 17025-accredited laboratory is generally accepted as adequate substantiation under FTC guidelines. The EU applies the same principle under the Common Criteria of EC 1223/2009.
Build Your Water Resistance Strategy with Vivify

Developing a water or sweat-resistant product that performs in testing and satisfies label requirements in both the U.S. and EU requires the right ingredient systems from the start. Vivify Beauty Care offers formulation support at every stage of development, with ingredient options for color cosmetics and sun care applications. For sunscreen filter selection context, review the organic vs. inorganic UV filters guide, or explore UV protection and defense actives. Ready to evaluate ingredients for your next formulation? Contact the Vivify team to discuss your project and request samples.
References
1. Pissavini, M., Pouradier, F., Wiener, G., Contier, M., Kolbe, L., Nogueira, L., Lapalud, P., Renner, G., & Matts, P. J. (2025). A new in vitro approach for determining the water resistance of sunscreen products, validated by a blinded ring test. International Journal of Cosmetic Science, 47(5). https://doi.org/10.1111/ics.13074
Disclaimer
The information provided in this blog is intended for general informational purposes only and is furnished without warranty, expressed or implied. The content reflects insights and information accurate to the best knowledge of Vivify Beauty Care at the time of publication.
This blog content should be used as a general guide and does not constitute a substitute for direct professional advice or product-specific consultation. Vivify Beauty Care does not validate any claims made within the blog, and customers bear the ultimate responsibility for ensuring their product applications and associated claims are compliant with all applicable laws and regulations. For specific inquiries or tailored recommendations regarding our product specifications and service offerings, please contact our sales professionals.



