Selecting between alpha hydroxy acids, beta hydroxy acids, and polyhydroxy acids is a formulation decision, not a performance preference. Each acid class brings distinct solubility profiles, effective pH windows, regulatory requirements by jurisdiction, and compatibility constraints that directly affect formulation design, processing parameters, and finished product safety. For R&D chemists and product development teams, AHA BHA PHA cosmetic formulation decisions shape the formula’s pH target, processing temperature, and which actives can coexist in the same product.
This guide is written for formulators, not consumers. It covers the chemistry basics you need as context, then moves directly into a practical decision framework: which acid type fits which product category, what pH windows and concentration ranges deliver activity, what the FDA and EU SCCS require, and which active combinations create problems in finished formulations.
Key Takeaways
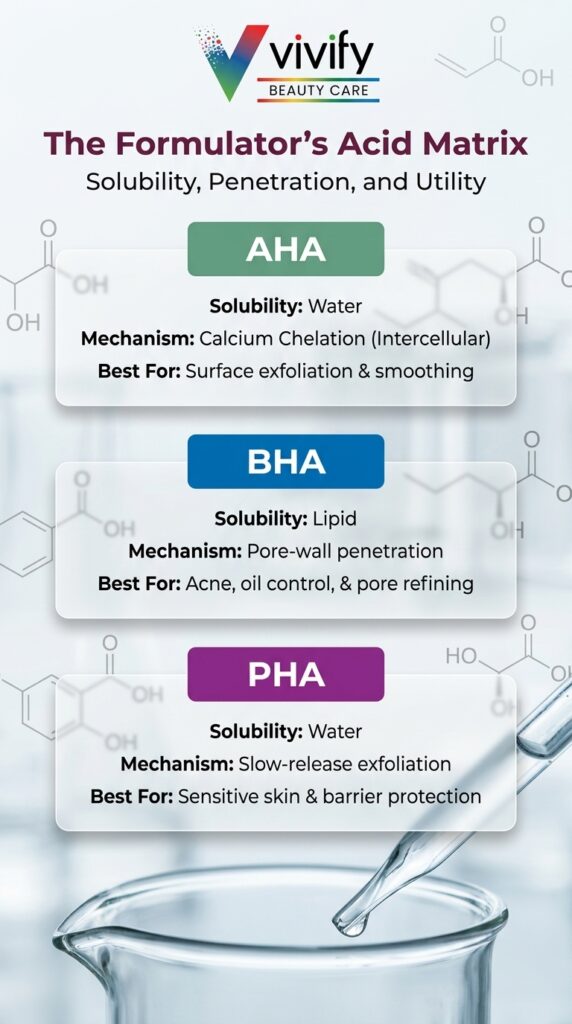
- AHAs are water-soluble and exfoliate via calcium chelation in the epidermis; optimal formulation pH is 3.5-4.5, with efficacy dropping substantially above pH 4.5.
- BHAs (primarily salicylic acid) are lipid-soluble and uniquely suited to follicular penetration; most effective at pH 3.0-4.0 at 0.5-2% in consumer cosmetics.
- PHAs such as gluconolactone and lactobionic acid penetrate more slowly than AHAs due to their larger molecular weight, delivering a gentler profile while adding antioxidant and barrier-enhancing properties.
- US FDA guidelines limit AHA consumer products to 10% or less at pH 3.5 or greater and require a sunburn alert statement on leave-on AHA formulations sold in the US.
- AHAs and BHAs at exfoliant concentrations are incompatible with peptides and high-load retinoids in the same product; separate these actives across different formulations.
- PHAs do not increase UV sensitivity as AHAs do and are well-documented as compatible with sensitive skin, rosacea, and post-procedure skin states.
AHA, BHA, and PHA: Core Chemistry for Formulators
AHAs are water-soluble organic acids that exfoliate by disrupting calcium-dependent cell adhesions in the stratum corneum. BHAs are lipid-soluble and penetrate the follicular canal, making them effective in sebum-rich environments. PHAs share the AHA exfoliant mechanism but carry larger molecular weights, which limits their penetration speed and significantly reduces irritation potential.

Alpha Hydroxy Acids (AHAs)
The AHA exfoliant mechanism operates through calcium chelation: reducing calcium ion availability in the epidermis disrupts the intercellular adhesions that hold cornified cells to the skin surface, triggering exfoliation.1 The major AHA actives in cosmetic formulation each carry distinct penetration and tolerability characteristics:
- Glycolic acid: smallest molecular weight in the class, allowing the deepest skin penetration; the standard benchmark for AHA efficacy
- Lactic acid: gentler profile with humectant properties from its structural similarity to the skin’s natural moisturizing factors; preferred when added hydration is part of the formulation goal
- Mandelic acid: larger molecular weight than glycolic and lactic; slower penetration suited to sensitive skin and brightening applications
- Malic and tartaric acid: primarily used as complementary actives and buffering agents in multi-acid formulations
Beta Hydroxy Acids (BHAs)
Beta hydroxy acids in cosmetic use are dominated by salicylic acid (SA). Technically, SA is an aromatic acid rather than a true beta hydroxy acid by strict organic chemistry nomenclature, but it is regulated and universally treated as a BHA by formulators and regulatory bodies.1 Its lipid solubility allows it to penetrate the follicular canal in ways water-soluble AHAs cannot, which is why it remains the primary acid choice for acne treatment and pore-refining formulations. Betaine salicylate offers a gentler BHA alternative that may be more appropriate for sensitive skin positioning.
Polyhydroxy Acids (PHAs)
Polyhydroxy acids, including gluconolactone and lactobionic acid, carry larger molecular weights than AHA molecules. This structural difference limits how quickly and deeply they penetrate the stratum corneum, which translates into a gentler clinical profile. PHAs also provide antioxidant and chelating properties that AHAs do not, adding formulation value beyond surface renewal.2
Formulation pH Windows and Processing Parameters
AHAs require a formulation pH of 3.5-4.5 for meaningful exfoliant activity. BHAs (salicylic acid) perform best at pH 3.0-4.0. PHAs are less pH-critical than AHAs for tolerability but still function best in mildly acidic conditions. For all three acid classes, raising pH to improve skin tolerance comes at a direct cost to exfoliant efficacy.
The table below summarizes effective pH ranges, consumer use level guidance, and key processing notes for each acid class:
| Acid Class | Effective pH Range | Consumer Use Level Guidance | Formulation Notes |
| AHA (glycolic, lactic, mandelic, malic, tartaric) | 3.5-4.5 | Up to 10% (US FDA OTC guidance); professional peel formulations up to approximately 20% | Heat-sensitive: add in cool-down phase (hot process) or use cold process. Avoid processing above 40°C. |
| BHA (salicylic acid; betaine salicylate as gentler alternative) | 3.0-4.0 | 0.5-2% in cosmetics; CIR safety assessment supports up to 3% | Lipid-soluble; requires solubilizer (propylene glycol or ethanol) for dispersion in aqueous formulas. |
| PHA (gluconolactone, lactobionic acid) | Mildly acidic; less pH-critical than AHAs | Refer to supplier technical data sheet for product-specific concentration guidance | More processing-flexible than AHAs; less temperature-sensitive; larger molecular weight allows broader formulation latitude. |
AHAs present a processing risk that BHAs and PHAs do not: heat sensitivity. In hot-process emulsification, add AHAs during the cool-down phase once the batch temperature drops below approximately 40°C. Cold process methods avoid this risk entirely and are preferred when the formulation stability profile allows.
Selecting the Right Exfoliant by Product Category
The most consequential variable in acid selection by product category is leave-on versus rinse-off contact time. AHAs and BHAs require sustained skin contact at a target pH to deliver meaningful exfoliant activity. Rinse-off applications can deliver results but require higher concentrations and formula optimization to compensate for reduced exposure time.
Leave-On Applications
Leave-on serums, treatment toners, and exfoliating pads are the primary delivery format for AHA and BHA actives. Extended contact time allows exfoliant activity at the concentration and pH levels where regulatory guidance is most clearly defined. Glycolic acid is the standard choice for maximum exfoliant effect; lactic acid is preferred when a gentler profile or added humectancy is the formulation goal.
For brightening-focused leave-on formulations, mandelic acid is frequently selected for its larger molecular weight, which slows penetration while still supporting surface brightness claims. Vivify’s brightening and skin lightening actives portfolio includes AHA options suited for hyperpigmentation and uneven tone formulations.
For BHA leave-on products targeting acne-prone or congested skin, salicylic acid at 0.5-2% in a pH 3.0-4.0 base delivers both surface exfoliation and follicular penetration. Vivify’s pore-refining actives portfolio includes BHA options for acne treatment and pore-refining leave-on formulations.
Rinse-Off Applications
Rinse-off AHA and BHA products require adequate concentration to deliver activity within a limited contact window. Cleansers and wash-off masks can carry AHAs up to the 10% US regulatory limit, but the effective exfoliant dose delivered to skin is lower than in leave-on formats because dilution and rinsing reduce active contact time. For rinse-off applications, glycolic acid is frequently used for its small molecular weight and well-established efficacy data.
BHA rinse-off formulations in cleansers targeting oily or acne-prone skin rely on salicylic acid’s ability to penetrate the follicular canal even during brief contact. Confirm that the base pH of the cleanser supports SA activity; many surfactant systems require pH adjustment to bring the finished formula into the 3.0-4.0 range needed for BHA performance. Vivify’s acne treatment actives portfolio supports both leave-on and rinse-off acid-containing formulations.
Sensitive Skin and Post-Procedure Formulations
PHAs are the preferred exfoliant class when the target is sensitive skin, rosacea-prone skin, or post-procedure recovery. Their larger molecular weight limits penetration speed, reducing the irritation, stinging, and erythema commonly associated with glycolic acid. The published clinical data on PHA tolerability in sensitive populations is direct:2
“PHAs have been found to be compatible with clinically sensitive skin, including rosacea and atopic dermatitis, and can be used after cosmetic procedures. PHAs provide additional humectant and moisturization properties compared with AHAs and can enhance stratum corneum barrier function, therefore increasing the skin’s resistance to chemical challenge.” 2
This makes gluconolactone and lactobionic acid strong candidates for post-peel aftercare products, barrier-repair serums with light exfoliant activity, and anti-aging formulations positioned for sensitive demographics. Explore Vivify’s polyhydroxy acid portfolio for PHA actives available for sampling and formulation consultation.
Regulatory Concentration Limits: FDA and EU SCCS

In the US, the FDA limits AHA consumer cosmetics to 10% or less at a pH of 3.5 or greater and requires a sunburn alert statement on leave-on AHA products. In the EU, glycolic acid is regulated as a restricted substance under Annex III of the EU Cosmetics Regulation; formulators developing products for EU distribution must confirm current limits with their regulatory compliance team.
The FDA’s AHA guidance, updated in November 2022, reflects adverse event monitoring: the agency recorded 114 AHA-related adverse event reports between 1992 and 2004, with the most serious reactions associated with high-concentration skin-peel products used outside professional settings. These data inform the 10% concentration and pH 3.5 minimum thresholds that apply to US consumer products. Professional peel formulations can use AHA concentrations up to approximately 20%, but concentrations above 10% typically require professional oversight across most regulatory jurisdictions.
BHA Concentration Guidance and EU Requirements
For BHAs, the CIR safety assessment supports use of salicylic acid in cosmetics at concentrations below 3%. The FDA’s guidance on beta hydroxy acids is maintained separately from the AHA guidance and should be reviewed alongside it when formulating products that contain both acid classes.
EU formulators face an additional layer of complexity. Glycolic acid is listed in Annex III of the EU Cosmetics Regulation as a restricted substance with specific conditions of use. Other AHAs, including lactic acid and mandelic acid, may carry different regulatory statuses under EU rules. Before finalizing concentrations for EU market formulations, confirm current SCCS guidance and Annex III listings with your regulatory compliance team.
Active Incompatibility: What to Avoid in Exfoliant Formulations
AHAs and BHAs at full exfoliant concentrations are not compatible with peptides or high-load retinoid formulations in the same product. The low-pH environment that enables exfoliant activity accelerates peptide hydrolysis and significantly amplifies the irritation, erythema, and flaking associated with retinoid actives. Both incompatibilities have practical formulation solutions.
AHAs, BHAs, and Peptides
Peptide stability is pH-dependent. At the pH required for exfoliant activity (3.0-4.5), peptide hydrolysis rates increase substantially. Formulating a peptide alongside AHAs or BHAs at exfoliant-effective concentrations risks degrading the peptide before the product reaches the consumer, undermining both efficacy and the claims the peptide was selected to support.
The practical solution is product separation: deliver the exfoliant and the peptide in different formulations. Alternatively, use delivery systems that maintain different local pH microenvironments for each ingredient. For a detailed guide on peptide stability, pH compatibility windows, and formulation design for peptide actives, see Vivify’s post on mastering peptides in skincare formulation.
AHAs, BHAs, and Retinoids
Combining strong AHA or BHA loads with retinoids (retinol, retinal, HPR, retinyl esters) in the same formulation significantly increases erythema, dryness, and flaking risk. Both ingredient classes accelerate cell turnover; the combination compounds the rate of exfoliation beyond what most skin barriers can tolerate. This concern intensifies at AHA concentrations above 5% and when the retinoid is a higher-potency form such as retinol or retinal rather than a retinyl ester.
The formulation solution is product separation. An exfoliant toner used in the morning and a retinoid serum used at night is a safer, better-tolerated system than combining both actives in a single formula. For formulation guidance on retinoid actives, including potency comparisons and compatibility considerations, see Vivify’s post on retinoid formulation.

Frequently Asked Questions About AHA BHA PHA Cosmetic Formulation
What is the effective pH range for AHA exfoliants?
AHAs are most effective at pH 3.5-4.5. Exfoliant activity declines significantly above pH 4.5 because the free acid form responsible for calcium chelation is reduced as pH rises.1 For US consumer products, pH 3.5 is the lower regulatory boundary per FDA guidance. Professional peel formulations can target lower pH values, but these are not appropriate for retail leave-on or rinse-off products.
Can AHA and BHA be formulated together in the same product?
Yes. AHA and BHA can be co-formulated provided the pH target, concentration limits, and product category support both actives. A common combination is glycolic or lactic acid with salicylic acid in an exfoliating toner at pH 3.5-4.0, which delivers surface renewal from the AHA and follicular clearance from the BHA.
This combination is appropriate for oily, acne-prone skin formulations. Manage total exfoliant load carefully to stay within FDA concentration guidance for the AHA component, and validate the formula’s irritation profile before launch.
Is salicylic acid technically a BHA?
Salicylic acid is classified and regulated as a BHA in cosmetics, but it is not a true beta hydroxy acid by strict organic chemistry nomenclature. Its hydroxy group sits on a benzene ring rather than at a beta-carbon position on an aliphatic chain, making it technically an aromatic acid.1 Despite this structural distinction, SA is universally treated as a BHA by formulators and regulatory agencies, and the classification is accurate for product development and regulatory compliance purposes.
Are PHAs suitable for sensitive skin and post-procedure formulations?
Yes. PHAs are well-documented as compatible with clinically sensitive skin, including rosacea and atopic dermatitis, and have been studied in post-cosmetic-procedure applications.2 Their larger molecular weight relative to AHAs limits penetration speed, reducing erythema and stinging. PHAs also support stratum corneum barrier function rather than compromising it, making them the lower-risk exfoliant choice for sensitive demographics or post-procedure aftercare product lines.
Do PHAs increase UV sensitivity the way AHAs do?
No. PHAs do not increase UV sensitivity. Glycolic acid has been shown to increase UV sensitivity by approximately 18% after four weeks of use.1 PHAs such as gluconolactone have demonstrated photoprotective properties in published research.2 This is a meaningful distinction for formulations intended for daytime use or populations with significant sun exposure. For AHA-containing leave-on products sold in the US, the FDA requires a sunburn alert statement on the product’s label.
Build Your Exfoliant Formulation with Vivify Beauty Care
Vivify Beauty Care offers a curated portfolio of AHA, BHA, and PHA actives to support formulation across product categories and skin targets. Explore the full exfoliant actives portfolio, or browse by acid class: AHA/BHA/PHA acids and beta hydroxy acids. For sample requests, formulation guidance, or technical questions about exfoliant actives, contact Vivify’s formulation team.
References
1. Kornhauser, A., Coelho, S. G., & Hearing, V. J. (2010). Applications of hydroxy acids: Classification, mechanisms, and photoactivity. Clinical, Cosmetic and Investigational Dermatology, 3, 135-142. https://doi.org/10.2147/CCID.S9042
2. Grimes, P. E., Green, B. A., Wildnauer, R. H., & Edison, B. L. (2004). The use of polyhydroxy acids (PHAs) in photoaged skin. Cutis, 73(2 Suppl), 3-13. PMID: 15002656. https://pubmed.ncbi.nlm.nih.gov/15002656/
Disclaimer
The information provided in this blog is intended for general informational purposes only and is furnished without warranty, expressed or implied. The content reflects insights and information accurate to the best knowledge of Vivify Beauty Care at the time of publication.
This blog content should be used as a general guide and does not constitute a substitute for direct professional advice or product-specific consultation. Vivify Beauty Care does not validate any claims made within the blog, and customers bear the ultimate responsibility for ensuring their product applications and associated claims are compliant with all applicable laws and regulations. For specific inquiries or tailored recommendations regarding our product specifications and service offerings, please contact our sales professionals.



